|
4/20/2024 0 Comments Atomic theory 
For example, if an element such as copper consists of only one kind of atom, then it cannot be broken down into simpler substances, that is, into substances composed of fewer types of atoms. 
Outline milestones in the development of modern atomic theory. Dalton’s atomic theory provides a microscopic explanation of the many macroscopic properties of matter that you’ve learned about. This module is an updated version of Atomic Theory I. The law of conservation of mass states that the total mass present before a chemical reaction is the same as the total mass present after the chemical reaction. Use postulates of Dalton’s atomic theory to explain the laws of definite and multiple proportions. atomic model, in physics, a model used to describe the structure and makeup of an atom. Atoms have protons and neutrons in the center, making the nucleus, while the electrons orbit the nucleus. Scientists did not account for the gases that play a critical role in this reaction. The atoms were unchangeable, indestructible, and always existed. 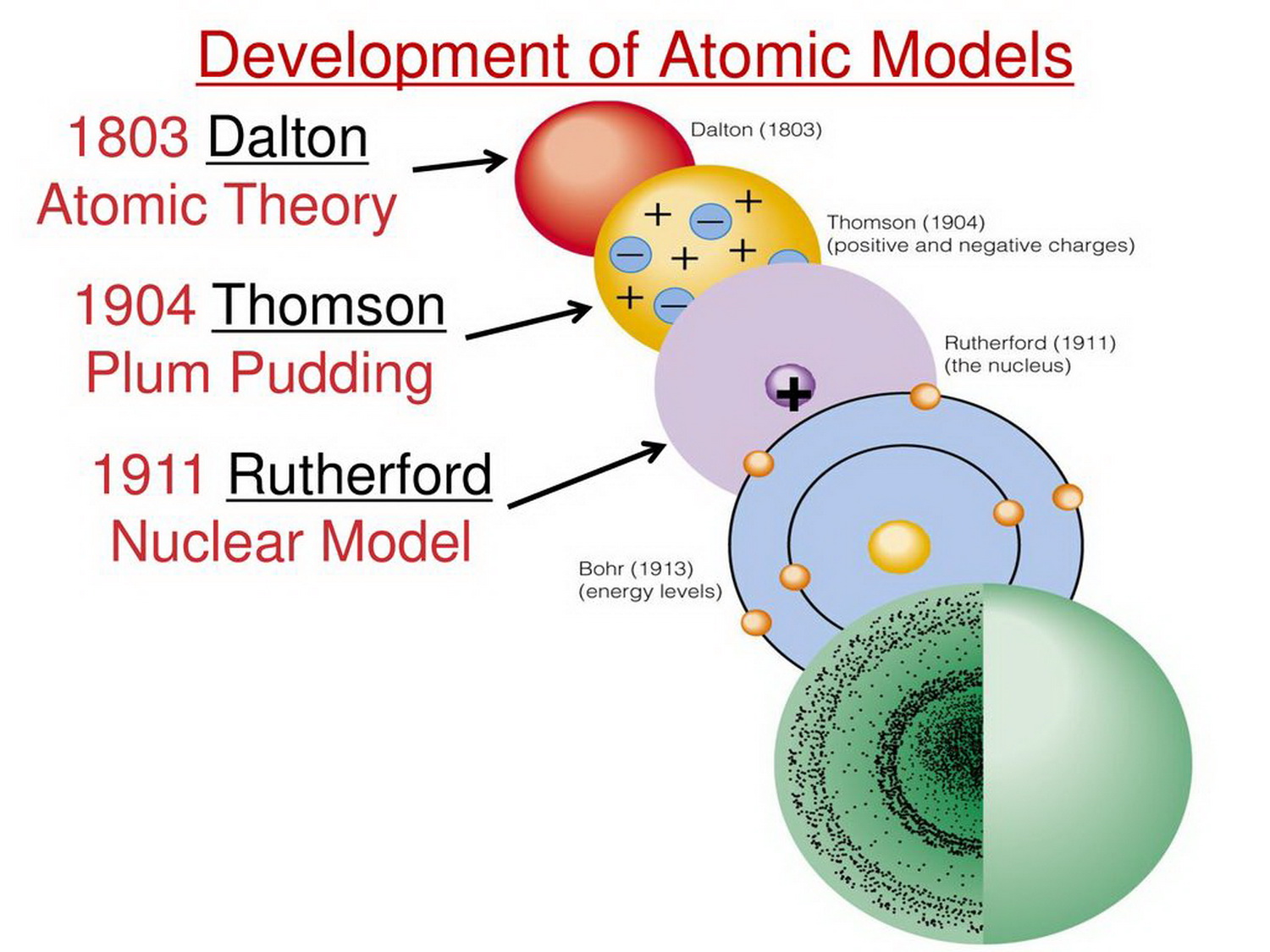
He taught that there were substances called atoms and that these atoms made up all material things. \) shows that the burning of word does follow the law of conservation of mass. One of these philosophers was Democritus (460-370 B.C.E.), often referred to as the 'laughing philosopher' because of his emphasis on cheerfulness. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
